Metal Plating
The basics of metal plating
Processes for Plating Metal
The several methods of metal plating include
Process of Electroplating
In the process of electroplating, a thin layer of metal is applied to the workpiece using electrodeposition. Engineers use controlled electrolysis to move the desired metal coating from the anode (the component containing the metal that will be used as the plating) to the cathode (the part that is to be plated). A constant electrical charge is provided while the cathode and anode are submerged in an electrolyte chemical bath.
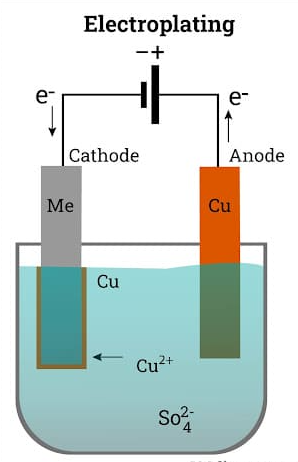
Positively charged ions (cations) flow to the cathode and negatively charged ions (anions) move to the anode as a result of the electric current, plating or producing a thin layer around the desired workpiece. A substrate material is taken and covered in a thin layer of metal, such as copper or nickel, by the electroplating process.
Due to the requirement that the substrate be made of a conductive material, electroplating is most frequently utilized on other metals. Different metals, most notably nickel and copper alloys, can be plated on plastic parts using other, less used autocatalytic pre-coating methods that create an incredibly thin conductive surface.
Between electroforming and electroplating
Electrodeposition is used in both electroplating and electroforming procedures. The usage of a mold in electroforming, which is discarded after forming a part, is the main distinction between these two methods.
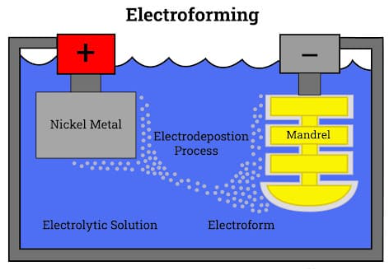
While electroplating is used to cover an existing workpiece made of a metal that is different from the original with a layer of metal, electroforming is used to create solid metal objects.
Components of electroplating materials
A combination of metals or a single metal can be electroplated onto an object. To boost the strength and conductivity, several manufacturers choose to layer metals like nickel and copper. The following is a list of the materials that are most frequently used in the electroplating process:
- Brass
- Cadmium
- Chromium
- Copper
- Gold
- Iron
- Nickel
- Silver
- Titanium
- Zinc
Substrates can be made from nearly any material, including plastic, stainless steel, and other sorts of metals. Organic materials that have been electroplated by artisans include soft fabric ribbons and flowers. It should be emphasized that non-conductive surfaces including plastic, glass, and wood need to be made conductive before going through the electroplating process. This can be done by applying a layer of conductive spray paint or paint on a non-conductive substrate.
Elemental Plating
The workpiece to be metal plated is immersed in a reducing agent that, when specific materials catalyze it, converts metal ions into metal that deposits around the workpiece. Electroless plating is a process of metal plating by chemical means rather than electrical means. The use of plastic materials, such as the plating of printed electronic circuits, has seen a significant expansion of the electroless plating technique. This procedure is used to metal plate many different consumer goods to provide beautiful and long-lasting surfaces.

The most popular electroless plating method is nickel plating, but copper, gold, and silver layers can also be deposited similarly. Other names for electroless plating include chemical plating and auto-catalytic plating.
A non-galvanic metal plating technique called electroless plating involves a number of simultaneous processes taking place in an aqueous solution. Without the usage of an external power source, the reactions take place.
A reducing agent, often sodium hypophosphite, releases hydrogen during the process and is then oxidized, leaving a negative charge on the part’s surface.
Electroless plating makes it possible to uniformly deposit metal over irregularly shaped objects, along edges, and within holes, which are difficult to metal plate by the electroplating process. It also allows the object to be bathed in a consistent metal ion concentration throughout.
The application of electroless plating is also done to deposit a conductive surface on a non-conductive object so that it can be electroplated with a metal coating.
